Abstract
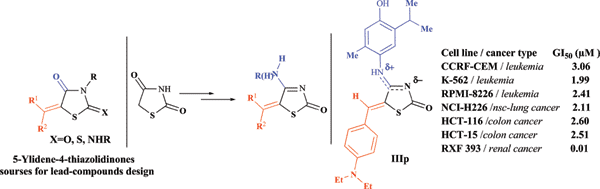
The synthesis and antitumor activity screening of 4-aminothiazol-2(5H)-one derivatives were performed. The absence of possible 4-amino-imino tautomerism of thiazolidinones-2 has been confirmed based on the study of the molecule structures. The existence of the alone amino-form was confirmed. An anticancer activity screening was performed within the Developmental Therapeutics Program (National Cancer Institute/NIH, USA). Tested compounds possess low to moderate anticancer activity (average values - 60 cancer cell lines assay) with significant selective action on certain cancer cell lines (CCRF-CEM and RPMI-8226/leukemia, U251/CNS cancer, RFX 393/renal cancer, OVCAR/ovarian cancer etc.). The advantage of 5-ylidene-4-R-amino derivatives in comparison with compounds with free amino group was shown. Some structure-activity findings, the comparison of target compounds with isomeric 5-ylidene-2-imino(amino)thiazol-4(5H)-ones, as well as COMPARE analysis were described. Among the tested compounds (Z)-5-(furan-2-ylmethylidene)-4-(4-chlorophenylamino)thiazol-2(5H)-one (IIIk) and (Z)-5-(4-diethylaminophenylmethylidene)-4-(4-hydroxy-5-isopropyl-2-methylphenylamino)thiazol-2(5H)-one (IIIp) possessed the highest levels of activity.
Keywords: Anticancer activity, 4-(Imino)amino-2-thiazolidinone, synthesis, X-ray study.
Medicinal Chemistry
Title:Synthesis and Evaluation of Anticancer Activity of 5-Ylidene-4- Aminothiazol-2(5H)-one Derivatives
Volume: 11 Issue: 6
Author(s): Danylo Kaminskyy, Ivanna Subtel’na, Borys Zimenkovsky, Olexandr Karpenko, Andrzej Gzella and Roman Lesyk
Affiliation:
Keywords: Anticancer activity, 4-(Imino)amino-2-thiazolidinone, synthesis, X-ray study.
Abstract: The synthesis and antitumor activity screening of 4-aminothiazol-2(5H)-one derivatives were performed. The absence of possible 4-amino-imino tautomerism of thiazolidinones-2 has been confirmed based on the study of the molecule structures. The existence of the alone amino-form was confirmed. An anticancer activity screening was performed within the Developmental Therapeutics Program (National Cancer Institute/NIH, USA). Tested compounds possess low to moderate anticancer activity (average values - 60 cancer cell lines assay) with significant selective action on certain cancer cell lines (CCRF-CEM and RPMI-8226/leukemia, U251/CNS cancer, RFX 393/renal cancer, OVCAR/ovarian cancer etc.). The advantage of 5-ylidene-4-R-amino derivatives in comparison with compounds with free amino group was shown. Some structure-activity findings, the comparison of target compounds with isomeric 5-ylidene-2-imino(amino)thiazol-4(5H)-ones, as well as COMPARE analysis were described. Among the tested compounds (Z)-5-(furan-2-ylmethylidene)-4-(4-chlorophenylamino)thiazol-2(5H)-one (IIIk) and (Z)-5-(4-diethylaminophenylmethylidene)-4-(4-hydroxy-5-isopropyl-2-methylphenylamino)thiazol-2(5H)-one (IIIp) possessed the highest levels of activity.
Export Options
About this article
Cite this article as:
Kaminskyy Danylo, Subtel’na Ivanna, Zimenkovsky Borys, Karpenko Olexandr, Gzella Andrzej and Lesyk Roman, Synthesis and Evaluation of Anticancer Activity of 5-Ylidene-4- Aminothiazol-2(5H)-one Derivatives, Medicinal Chemistry 2015; 11 (6) . https://dx.doi.org/10.2174/1573406411666150211112049
| DOI https://dx.doi.org/10.2174/1573406411666150211112049 |
Print ISSN 1573-4064 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1875-6638 |
Call for Papers in Thematic Issues
Carbohydrates in Computational and Medicinal Chemistry
Carbohydrates are the most essential organic molecules and are involved in the maintenance of various physiological and metabolic processes in living organisms. Carbohydrate-based compounds have come to the attention of researchers because of their significant contributions to biological functions, such as cell development and cell proliferation, connections between several cells, ...read more
Recent Advances in the Medicinal Chemistry of Cancer
Scope of the Thematic Issue: Correlation between structure and function is one of the important aspects of the success of anti-cancer compounds associated with their structure-activity interactions, physiology, biochemical, molecular, and genetic processes. Overcoming these obstacles is key to obtaining further insights into developments in rational drug design, bioorganic chemistry, ...read more
 36
36 1
1 1
1
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
Related Articles
-
A Comparison of Physicochemical Property Profiles of Marketed Oral Drugs and Orally Bioavailable Anti-Cancer Protein Kinase Inhibitors in Clinical Development
Current Topics in Medicinal Chemistry Nanoparticles in Biomedicine: New Insights from Plant Viruses
Current Medicinal Chemistry Exploiting Cyclooxygenase-(in)Dependent Properties of COX-2 Inhibitors for Malignant Glioma Therapy
Anti-Cancer Agents in Medicinal Chemistry Towards Understanding the Roles of Prohibitins, Multi-Functional Regulator Proteins
Current Chemical Biology Histone Modifier Differentially Regulates Gene Expression and Unravels Survival Role of MicroRNA-494 in Jurkat Leukemia
MicroRNA A Role for Tumor Suppressor Protein p53 in the Fidelity of DNA Synthesis and Resistance Towards Nucleoside Analogs
Current Cancer Therapy Reviews Expression, Purification, Crystallization and Preliminary X-Ray Analysis of Human Spindlin1, an Ovarian Cancer-Related Protein
Protein & Peptide Letters Potential Non-coding RNAs from Microorganisms and their Therapeutic Use in the Treatment of Different Human Cancers
Current Gene Therapy Effects of Iron Chelation in Osteosarcoma
Current Cancer Drug Targets The Clinical Prognostic Value of LRG1 in Esophageal Squamous Cell Carcinoma
Current Cancer Drug Targets Targeting the Cancer-Stroma Interaction: A Potential Approach for Pancreatic Cancer Treatment
Current Pharmaceutical Design Receptor Mediated Tumor Targeting: An Emerging Approach for Cancer Therapy
Current Drug Delivery Tumor Initiating Cells
Current Pharmaceutical Biotechnology Role of Rap2 and its Downstream Effectors in Tumorigenesis
Anti-Cancer Agents in Medicinal Chemistry MiR-138 and MiR-135 Directly Target Focal Adhesion Kinase, Inhibit Cell Invasion, and Increase Sensitivity to Chemotherapy in Cancer Cells
Anti-Cancer Agents in Medicinal Chemistry Copper Complexes of 8-Aminoquinoline and Uracils as Novel Aromatase Inhibitors
Letters in Drug Design & Discovery The Urokinase Receptor Interactome
Current Pharmaceutical Design Light Directed Gene Transfer by Photochemical Internalisation
Current Gene Therapy The Weal and Woe of Costimulation in the Adoptive Therapy of Cancer with Chimeric Antigen Receptor (CAR)-Redirected T Cells
Current Molecular Medicine "Take and Run" and "Take and Hold" Therapeutical Electromagnetic Field Application Modalities in Physiotherapy: Some Clinically Significant Pathways of Action
Current Chemical Biology