Abstract
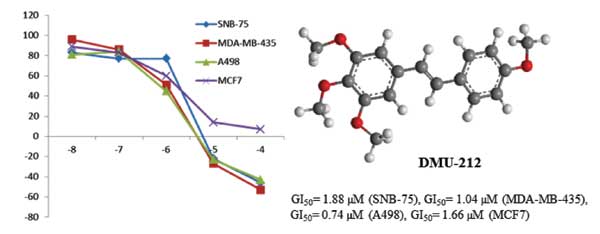
DMU-212, a methoxylated resveratrol analog, has significant anticancer activity, and selectively targets tumor cells. A library of E-diarylstilbenes structurally related to DMU-212 has been synthesized and evaluated for anticancer activity against a large panel of 45 human cancer cell lines. From this study, DMU-212 (3a) exhibited an average growth inhibitory effect (GI50) of 3.5 M against all the human cancer cell lines in the panel, and was particularly effective against the four cancer cell lines: SNB-75 (CNS), MDA-MB-435 (melanoma), A498 (renal), and MCF7 (breast), with GI50 values of 1.88, 1.04, 0.74 and 1.66 µM, respectively. Also, the 4’-chloro analog of DMU-212, 3d, exhibited 98 and 80 percent growth inhibition against MDA-MB-435 (melanoma) and K-562 (leukemia) cancer cell lines at a concentration of 10 M. Further investigation of DMU-212 and its analogs may provide novel therapeutic avenues for treatment of a variety of human cancers.
Keywords: Resveratrol, DMU-212, anticancer, growth inhibition, cytotoxicity, diarylstilbenes.
Letters in Drug Design & Discovery
Title:Anticancer Evaluation of 3,4,5,4'-trans-tetramethoxystilbene (DMU-212) and Its Analogs Against an Extensive Panel of Human Tumor Cell Lines
Volume: 12 Issue: 7
Author(s): Nikhil Reddy Madadi and Peter Anthony Crooks
Affiliation:
Keywords: Resveratrol, DMU-212, anticancer, growth inhibition, cytotoxicity, diarylstilbenes.
Abstract: DMU-212, a methoxylated resveratrol analog, has significant anticancer activity, and selectively targets tumor cells. A library of E-diarylstilbenes structurally related to DMU-212 has been synthesized and evaluated for anticancer activity against a large panel of 45 human cancer cell lines. From this study, DMU-212 (3a) exhibited an average growth inhibitory effect (GI50) of 3.5 M against all the human cancer cell lines in the panel, and was particularly effective against the four cancer cell lines: SNB-75 (CNS), MDA-MB-435 (melanoma), A498 (renal), and MCF7 (breast), with GI50 values of 1.88, 1.04, 0.74 and 1.66 µM, respectively. Also, the 4’-chloro analog of DMU-212, 3d, exhibited 98 and 80 percent growth inhibition against MDA-MB-435 (melanoma) and K-562 (leukemia) cancer cell lines at a concentration of 10 M. Further investigation of DMU-212 and its analogs may provide novel therapeutic avenues for treatment of a variety of human cancers.
Export Options
About this article
Cite this article as:
Madadi Reddy Nikhil and Crooks Anthony Peter, Anticancer Evaluation of 3,4,5,4'-trans-tetramethoxystilbene (DMU-212) and Its Analogs Against an Extensive Panel of Human Tumor Cell Lines, Letters in Drug Design & Discovery 2015; 12 (7) . https://dx.doi.org/10.2174/1570180812999150324163710
| DOI https://dx.doi.org/10.2174/1570180812999150324163710 |
Print ISSN 1570-1808 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1875-628X |
 31
31 2
2
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
Related Articles
-
Recent Advances in Heterocyclic Tubulin Inhibitors Targeting the Colchicine Binding Site
Anti-Cancer Agents in Medicinal Chemistry Melittin: A Natural Peptide with Expanded Therapeutic Applications
The Natural Products Journal Ceramide and Apoptosis: Exploring the Enigmatic Connections between Sphingolipid Metabolism and Programmed Cell Death
Anti-Cancer Agents in Medicinal Chemistry Feud or Friend? The Role of the miR-17-92 Cluster in Tumorigenesis
Current Genomics Endothelial Dysfunction in Metabolic Diseases: Role of Oxidation and Possible Therapeutic Employment of N-acetylcysteine
Current Medicinal Chemistry Identification of Novel Structurally Diverse Anaplastic Lymphoma Kinase Inhibitors Based on Pharmacophore Modeling, Virtual Screening and Molecular Docking
Combinatorial Chemistry & High Throughput Screening Evaluation of the Anticancer Activities of the Plant Alkaloids Sanguinarine and Chelerythrine in Human Breast Adenocarcinoma Cells
Anti-Cancer Agents in Medicinal Chemistry Current Review of Small Molecule Ret Kinase Inhibitors
Mini-Reviews in Medicinal Chemistry Namitecan: a Hydrophilic Camptothecin with a Promising Preclinical Profile
Current Medicinal Chemistry Bioactive N-Phenylimidazole Derivatives
Current Chemical Biology Recent Progress of Src SH2 and SH3 Inhibitors as Anticancer Agents
Current Medicinal Chemistry Pharmacological Activation of p53 in Cancer Cells
Current Pharmaceutical Design Plant Polyphenols and Tumors: From Mechanisms to Therapies, Prevention, and Protection Against Toxicity of Anti-Cancer Treatments
Current Medicinal Chemistry Plant Bioactive Peptides: Current Status and Prospects Towards Use on Human Health
Protein & Peptide Letters Genetics and Bioenergetics of Mitochondria Influencing the Etiology and Pharmacology of Steroidal Hormones
Current Pharmacogenomics The Importance of Melatonin and Mitochondria Interaction in Mood Disorders and Schizophrenia: A Current Assessment
Current Medicinal Chemistry Polyamine Modulation of NMDARs as a Mechanism to Reduce Effects of Alcohol Dependence
Recent Patents on CNS Drug Discovery (Discontinued) Induced Pluripotent Stem Cell-Based Studies of Parkinson's Disease: Challenges and Promises
CNS & Neurological Disorders - Drug Targets ANTI-ADHESION Evolves To a Promising Therapeutic Concept in Oncology
Current Medicinal Chemistry Peptide-Drug Conjugate: A Novel Drug Design Approach
Current Medicinal Chemistry