Abstract
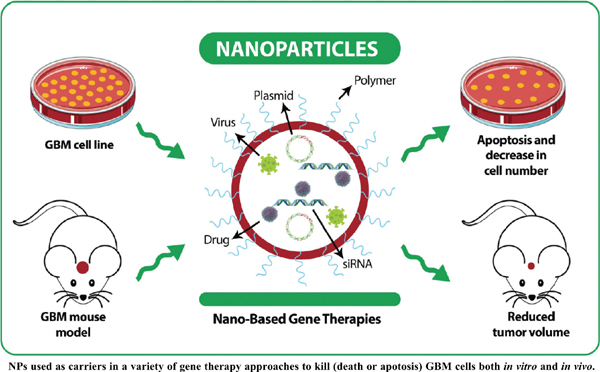
Background: Glioblastoma multiforme (GBM) is the most common and fatal type of glioma. Nanoparticles (NPs) are used in new approaches for the delivery of gene therapy in the treatment of GBM.
Introduction: The purpose of this article was to review the efficacy of NPs as the targeted carriers in the gene therapy aimed at apoptosis in GBM.
Methods: The appropriate keywords such as nanoparticle, glioblastoma, gene therapy, apoptosis, and related words were used to search from PubMed, ISI Web of Science, and Scopus for relevant publications up to September 4, 2020, with no language restrictions. The present systematic review was performed based on PRISMA protocol and reviewed the articles evaluating the effects of nanoparticles, carriers of various gene therapies essentials, on GBM cells apoptosis in vitro and in vivo. The selected articles were considered using specific scores on the quality of the articles. Data extraction and quality evaluation were performed by two reviewers.
Results: Of 101 articles retrieved, forty-two met the inclusion criteria and were, therefore, subjected to the final deduction. The most widely used NP in GBM gene therapy studies is polyamidoamine (PAMAM). The most common gene therapy approach for apoptosis in GBM is using siRNAs.
Conclusion: In conclusion, these studies validated that NPs could be a practical choice to enhance the efficiency and specific delivery in gene therapies for GBM cell apoptosis. However, the choice of NP type and gene therapy mechanism affect the GBM cell apoptotic efficiency.
Keywords: Nanoparticle, gene therapy, glioblastoma, apoptosis, PRISMA, polyamidoamine.
[http://dx.doi.org/10.3390/nano9010105] [PMID: 30654536]
[http://dx.doi.org/10.1593/neo.04535] [PMID: 15720813]
[http://dx.doi.org/10.1093/carcin/bgh283] [PMID: 15375012]
[http://dx.doi.org/10.4172/2161-0444.1000247]
[http://dx.doi.org/10.1016/j.jsps.2017.10.012] [PMID: 29379334]
[http://dx.doi.org/10.1371/journal.pmed.1000100] [PMID: 19621070]
[http://dx.doi.org/10.3390/pharmaceutics10040282] [PMID: 30558378]
[http://dx.doi.org/10.3390/biomedicines4030020] [PMID: 28536387]
[http://dx.doi.org/10.2147/IJN.S164364] [PMID: 30214189]
[http://dx.doi.org/10.2147/IJN.S189871] [PMID: 31043779]
[http://dx.doi.org/10.1016/j.jconrel.2016.08.029] [PMID: 27569663]
[http://dx.doi.org/10.2147/DDDT.S95843] [PMID: 26719669]
[http://dx.doi.org/10.1021/nn504905q] [PMID: 25643235]
[http://dx.doi.org/10.1016/j.biomaterials.2011.05.064] [PMID: 21700333]
[http://dx.doi.org/10.1039/C6CS00636A] [PMID: 28585944]
[http://dx.doi.org/10.2147/IJN.S146315]
[http://dx.doi.org/10.4103/0971-6866.112870] [PMID: 23901186]
[http://dx.doi.org/10.1002/jgm.2698] [PMID: 23355455]
[http://dx.doi.org/10.2174/1566523219666190719100526] [PMID: 31566126]
[http://dx.doi.org/10.1080/21691401.2018.1500372]
[http://dx.doi.org/10.1007/s11051-014-2342-1]
[http://dx.doi.org/10.2174/1566523220666201012150130] [PMID: 33045966]
[http://dx.doi.org/10.14740/jocmr3781] [PMID: 31143308]
[http://dx.doi.org/10.1016/j.biotechadv.2019.107502] [PMID: 31887345]
[http://dx.doi.org/10.1007/s12250-019-00121-4] [PMID: 31218589]
[http://dx.doi.org/10.2147/IJN.S148359] [PMID: 29238188]
[http://dx.doi.org/10.1016/j.jconrel.2019.01.034] [PMID: 30690106]
[http://dx.doi.org/10.1016/j.biomaterials.2019.04.020] [PMID: 31026613]
[http://dx.doi.org/10.3389/fimmu.2018.00824] [PMID: 29740437]
[http://dx.doi.org/10.1080/21691401.2018.1489824] [PMID: 30092165]
[http://dx.doi.org/10.1002/advs.201600491] [PMID: 28852611]
[http://dx.doi.org/10.1016/j.ymthe.2017.04.023] [PMID: 28502470]
[http://dx.doi.org/10.1016/j.ymthe.2016.11.016] [PMID: 28109960]
[http://dx.doi.org/10.1016/j.nano.2017.06.004] [PMID: 28614736]
[http://dx.doi.org/10.1038/ncomms15144] [PMID: 28489075]
[http://dx.doi.org/10.2174/1566523217666170605115829] [PMID: 28578643]
[http://dx.doi.org/10.1016/j.xphs.2017.01.034] [PMID: 28188727]
[http://dx.doi.org/10.1002/btm2.10019] [PMID: 28349127]
[http://dx.doi.org/10.1016/j.jconrel.2016.10.027] [PMID: 27794494]
[http://dx.doi.org/10.1016/j.ijpharm.2016.09.083] [PMID: 27732896]
[http://dx.doi.org/10.1016/j.biomaterials.2014.10.034] [PMID: 25453963]
[http://dx.doi.org/10.1039/C5RA18464A]
[http://dx.doi.org/10.1002/adhm.201500563] [PMID: 26498165]
[http://dx.doi.org/10.1186/s13287-015-0157-3] [PMID: 26345383]
[http://dx.doi.org/10.1016/j.jconrel.2015.04.002] [PMID: 25861727]
[http://dx.doi.org/10.1021/nn506248s] [PMID: 25558928]
[http://dx.doi.org/10.1117/1.JBO.19.10.105009] [PMID: 25341069]
[http://dx.doi.org/10.1038/mtna.2014.37] [PMID: 25118171]
[http://dx.doi.org/10.2217/nnm.14.12]
[http://dx.doi.org/10.1021/nn5014484] [PMID: 24811110]
[http://dx.doi.org/10.1016/j.biomaterials.2014.07.043] [PMID: 25112935]
[http://dx.doi.org/10.1016/j.biomaterials.2013.02.067] [PMID: 23522375]
[http://dx.doi.org/10.1016/j.biomaterials.2013.08.030] [PMID: 23993342]
[http://dx.doi.org/10.1126/scitranslmed.3006839] [PMID: 24174328]
[http://dx.doi.org/10.1016/j.jconrel.2013.08.020] [PMID: 24008150]
[http://dx.doi.org/10.1016/j.ijpharm.2013.01.057] [PMID: 23384727]
[http://dx.doi.org/10.2147/IJN.S39072] [PMID: 23589689]
[http://dx.doi.org/10.1021/mp3002039] [PMID: 22853427]
[http://dx.doi.org/10.1016/j.biomaterials.2010.11.079] [PMID: 21185076]
[http://dx.doi.org/10.1002/jbm.a.32525] [PMID: 19591231]
[http://dx.doi.org/10.1158/0008-5472.CAN-06-2354] [PMID: 17178885]
[http://dx.doi.org/10.1038/sj.cgt.7700357] [PMID: 11593333]
[http://dx.doi.org/10.1038/s41598-017-01279-1] [PMID: 28450700]
[http://dx.doi.org/10.3760/cma.j.issn.0253-3766.2012.05.002] [PMID: 22883450]