Abstract
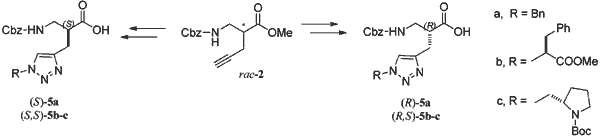
In recent years, peptidomimetics have gained enormous importance in drug design aiming to achieve increased drug metabolic stability and higher selectivity. In the field of peptidomimetics, β-peptides incorporating β2- and β3-amino acids (the higher homologs of natural α-amino acids) provide a powerful method for the synthesis of peptidomimetics with particular secondary structures. In this regard, 1,2,3-triazole-modified peptidomimetics can act as effective peptide surrogates, and therefore have gained considerable attention. In the present report, 1,4-disubstituted 1,2,3-triazoles attached to β-amino acids were prepared selectively from the corresponding alkynyl-β2-amino acids according to Huisgen’s copper-catalyzed 1,3-dipolar cycloaddition (CuAAC), under mild conditions and with very high efficiency. Different azide derivatives, including some incorporating α-amino acids, were employed in this cycloaddition reaction. The enantiopure compounds were obtained via diastereomeric salt formation with chiral adjuvants, and subsequent separation.
Keywords: Triazol, click-reaction, β-amino acids, peptidomimetics, enantio- and diastereoseparation.
Current Topics in Medicinal Chemistry
Title:Enantiopure 1,2,3-Triazolyl-β-amino Acids via Click Cycloaddition Reaction on Racemic Alkynyl Precursors Followed by Separation of Stereoisomers
Volume: 14 Issue: 10
Author(s): Margarita Escudero-Casao, Alberto Vega-Penaloza and Eusebio Juaristi
Affiliation:
Keywords: Triazol, click-reaction, β-amino acids, peptidomimetics, enantio- and diastereoseparation.
Abstract: In recent years, peptidomimetics have gained enormous importance in drug design aiming to achieve increased drug metabolic stability and higher selectivity. In the field of peptidomimetics, β-peptides incorporating β2- and β3-amino acids (the higher homologs of natural α-amino acids) provide a powerful method for the synthesis of peptidomimetics with particular secondary structures. In this regard, 1,2,3-triazole-modified peptidomimetics can act as effective peptide surrogates, and therefore have gained considerable attention. In the present report, 1,4-disubstituted 1,2,3-triazoles attached to β-amino acids were prepared selectively from the corresponding alkynyl-β2-amino acids according to Huisgen’s copper-catalyzed 1,3-dipolar cycloaddition (CuAAC), under mild conditions and with very high efficiency. Different azide derivatives, including some incorporating α-amino acids, were employed in this cycloaddition reaction. The enantiopure compounds were obtained via diastereomeric salt formation with chiral adjuvants, and subsequent separation.
Export Options
About this article
Cite this article as:
Escudero-Casao Margarita, Vega-Penaloza Alberto and Juaristi Eusebio, Enantiopure 1,2,3-Triazolyl-β-amino Acids via Click Cycloaddition Reaction on Racemic Alkynyl Precursors Followed by Separation of Stereoisomers, Current Topics in Medicinal Chemistry 2014; 14 (10) . https://dx.doi.org/10.2174/1568026614666140423110420
| DOI https://dx.doi.org/10.2174/1568026614666140423110420 |
Print ISSN 1568-0266 |
| Publisher Name Bentham Science Publisher |
Online ISSN 1873-4294 |
Call for Papers in Thematic Issues
Chemistry Based on Natural Products for Therapeutic Purposes
The development of new pharmaceuticals for a wide range of medical conditions has long relied on the identification of promising natural products (NPs). There are over sixty percent of cancer, infectious illness, and CNS disease medications that include an NP pharmacophore, according to the Food and Drug Administration. Since NP ...read more
Current Trends in Drug Discovery Based on Artificial Intelligence and Computer-Aided Drug Design
Drug development discovery has faced several challenges over the years. In fact, the evolution of classical approaches to modern methods using computational methods, or Computer-Aided Drug Design (CADD), has shown promising and essential results in any drug discovery campaign. Among these methods, molecular docking is one of the most notable ...read more
Drug Discovery in the Age of Artificial Intelligence
In the age of artificial intelligence (AI), we have witnessed a significant boom in AI techniques for drug discovery. AI techniques are increasingly integrated and accelerating the drug discovery process. These developments have not only attracted the attention of academia and industry but also raised important questions regarding the selection ...read more
From Biodiversity to Chemical Diversity: Focus of Flavonoids
Flavonoids are the largest group of polyphenols, plant secondary metabolites arising from the essential aromatic amino acid phenylalanine (or more rarely from tyrosine) via the phenylpropanoid pathway. The flavan nucleus is the basic 15-carbon skeleton of flavonoids (C6-C3-C6), which consists of two phenyl rings (A and B) and a heterocyclic ...read more
 37
37
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
- Announcements
Related Articles
-
A Retrospective Study on the Incidence of Seizures among Neurosurgical Patients Who Treated with Imipenem/Cilastatin or Meropenem
Current Pharmaceutical Biotechnology Folate Status and Depressive Symptoms in Reproductive-Age Women
Current Nutrition & Food Science Effects of Chronic Lamotrigine Administration on Maximal Electroshock- Induced Seizures in Mice
CNS & Neurological Disorders - Drug Targets Psychoemotional Background of Temporomandibular Joint Dysfunction and Possible Drug Therapy
Letters in Drug Design & Discovery Neurocysticercosis: The Enigmatic Disease
Central Nervous System Agents in Medicinal Chemistry Aligning Animal Models of Clinical Germinal Matrix Hemorrhage, From Basic Correlation to Therapeutic Approach
Current Drug Targets Predicting Targeted Polypharmacology for Drug Repositioning and Multi- Target Drug Discovery
Current Medicinal Chemistry Developmental Expression and Dysregulation of miR-146a and miR-155 in Down's Syndrome and Mouse Models of Down's Syndrome and Alzheimer's Disease
Current Alzheimer Research Signaling Pathways Involved in Antidepressant-Induced Cell Proliferation and Synaptic Plasticity
Current Pharmaceutical Design Minocycline and Doxycycline: More Than Antibiotics
Current Molecular Pharmacology ATP-Binding Cassette Transporters at the Blood-Brain Barrier in Ischaemic Stroke
Current Pharmaceutical Design Pleiotropic, Cardioprotective Effects of Omega-3 Polyunsaturated Fatty Acids
Mini-Reviews in Medicinal Chemistry Electroencephalography and Dementia: A Literature Review and Future Perspectives
CNS & Neurological Disorders - Drug Targets Implications of Prion Protein Biology
Current Neurovascular Research Role of Resveratrol and its Analogues in the Treatment of Neurodegenerative Diseases: Focus on Recent Discoveries
CNS & Neurological Disorders - Drug Targets Retraction Notice: Potential Application of Venom Proteins in Designing of Medicines for Treating Human Neurodegenerative Disorders
Protein & Peptide Letters An Insight into Drug Repositioning for the Development of Novel Anti-Cancer Drugs
Current Topics in Medicinal Chemistry Recent Advances in the Treatment of Neurogenic Erectile Dysfunction
Recent Patents on CNS Drug Discovery (Discontinued) Pharmacology of Voltage-Gated Proton Channels
Current Pharmaceutical Design The Treatment of Painful Diabetic Neuropathy
Current Diabetes Reviews