Abstract
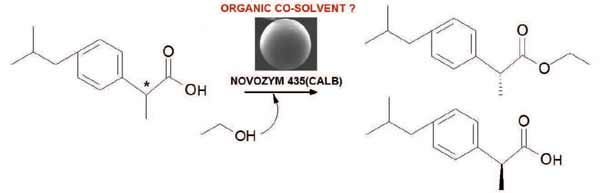
The commercial biocatalyst Novozym ® 435 was used for the kinetic resolution of (R/S)-ibuprofen through the esterification with short chain alcohols (ethanol, 1-propanol and 2-propanol) in the absence of organic co-solvent. The best enzymatic performance was obtained by employing ethanol as reagent and solvent. Due to the deleterious effect of this alcohol on the integrity of the commercial biocatalyst previously reported different organic co-solvents (isooctane, nhexane, carbon tetrachloride, ethyl acetate, acetonitrile and tetrahydrofuran) were screened in order to minimize the volume of ethanol to be used. Thus, the effect of the chemical nature of the co-solvent on enantioselective esterification of (R/S)-ibuprofen with ethanol was evaluated. The results show that the best performance was obtained with the reaction system without co-solvent added. Additionally, this investigation demonstrated the need to address multiple physicochemical properties of the solvents to analyze their effects on biocatalysis.
Keywords: Ibuprofen, Novozym435, Ethanol, Organic solvents.
Current Catalysis
Title:Effect of Co-solvents in the Enantioselective Esterification of (R/S)- ibuprofen with Ethanol
Volume: 3 Issue: 2
Author(s): Carla Jose, M. Victoria Toledo, Jaiver Osorio Grisales and Laura E. Briand
Affiliation:
Keywords: Ibuprofen, Novozym435, Ethanol, Organic solvents.
Abstract: The commercial biocatalyst Novozym ® 435 was used for the kinetic resolution of (R/S)-ibuprofen through the esterification with short chain alcohols (ethanol, 1-propanol and 2-propanol) in the absence of organic co-solvent. The best enzymatic performance was obtained by employing ethanol as reagent and solvent. Due to the deleterious effect of this alcohol on the integrity of the commercial biocatalyst previously reported different organic co-solvents (isooctane, nhexane, carbon tetrachloride, ethyl acetate, acetonitrile and tetrahydrofuran) were screened in order to minimize the volume of ethanol to be used. Thus, the effect of the chemical nature of the co-solvent on enantioselective esterification of (R/S)-ibuprofen with ethanol was evaluated. The results show that the best performance was obtained with the reaction system without co-solvent added. Additionally, this investigation demonstrated the need to address multiple physicochemical properties of the solvents to analyze their effects on biocatalysis.
Export Options
About this article
Cite this article as:
Jose Carla, Toledo Victoria M., Grisales Osorio Jaiver and Briand E. Laura, Effect of Co-solvents in the Enantioselective Esterification of (R/S)- ibuprofen with Ethanol, Current Catalysis 2014; 3 (2) . https://dx.doi.org/10.2174/2211544702666131230234058
| DOI https://dx.doi.org/10.2174/2211544702666131230234058 |
Print ISSN 2211-5447 |
| Publisher Name Bentham Science Publisher |
Online ISSN 2211-5455 |
 30
30
- Author Guidelines
- Graphical Abstracts
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Publishing Ethics and Rectitude
- Increase Visibility of Your Article
- Archiving Policies
- Peer Review Workflow
- Order Your Article Before Print
- Promote Your Article
- Manuscript Transfer Facility
- Editorial Policies
- Allegations from Whistleblowers
- Announcements
Related Articles
-
Promises of Nanotechnology for Drug Delivery to Brain in Neurodegenerative Diseases
Current Nanoscience Heterogeneous Diastereoselective Catalysis - A Powerful Strategy Toward C(15) Stereoselectivity from PGF<sub>2α</sub> Analogues Structure
Current Pharmaceutical Design Moxifloxacin Hydrochloride-Loaded Eudragit® RL 100 and Kollidon® SR Based Nanoparticles: Formulation, In vitro Characterization and Cytotoxicity
Combinatorial Chemistry & High Throughput Screening The Central Vasopressinergic System: Examining the Opportunities for Psychiatric Drug Development
Current Pharmaceutical Design Naturally Occurring and Synthetic Bioactive Molecules as Novel Non-Nucleoside HBV Inhibitors
Mini-Reviews in Medicinal Chemistry Immunomodulatory Effects of Vitamin D in Influenza Infection
Current Immunology Reviews (Discontinued) What We have Learned about Pain from Rodent Models of Arthritis?
Current Rheumatology Reviews A Review of Recent Patents on the ASICs as a Key Drug Target
Recent Patents on Biotechnology Germ Cell Apoptosis: Relevance to Infertility and Contraception
Immunology, Endocrine & Metabolic Agents in Medicinal Chemistry (Discontinued) Role of Retinoids, Rexinoids and Thyroid Hormone in the Expression of Cytochrome P450 Enzymes
Current Drug Metabolism Lectin Microarrays: A Powerful Tool for Glycan-Based Biomarker Discovery
Combinatorial Chemistry & High Throughput Screening Supplemented Infant Formulas: Which is the Best?
Current Nutrition & Food Science Foldable/Expandable Gastro-retentive Films Based on Starch and Chitosan as a Carrier For Prolonged Release of Resveratrol
Current Pharmaceutical Biotechnology Virus-Specific Peptide Dependent NK Cell Cytotoxicity
Inflammation & Allergy - Drug Targets (Discontinued) Reconstruction of the 1918 Pandemic Influenza Virus: How Revealing the Molecular Secrets of the Virus Responsible for the Worst Pandemic in Recorded History Can Guide Our Response to Future Influenza Pandemics
Infectious Disorders - Drug Targets Pharmacological Strategies to Overcome HER2 Cross-Talk and Trastuzumab Resistance
Current Medicinal Chemistry Oncolytic HSV-1 Virotherapy: Clinical Experience and Opportunities for Progress
Current Pharmaceutical Biotechnology Antimicrobial Proteins from Snake Venoms: Direct Bacterial Damage and Activation of Innate Immunity Against Staphylococcus aureus Skin Infection
Current Medicinal Chemistry Data Reduction Methods for Application of Fluorescence Correlation Spectroscopy to Pharmaceutical Drug Discovery
Current Pharmaceutical Biotechnology ETS Proteins and MMPs: Partners in Invasion and Metastasis
Current Drug Targets