Abstract
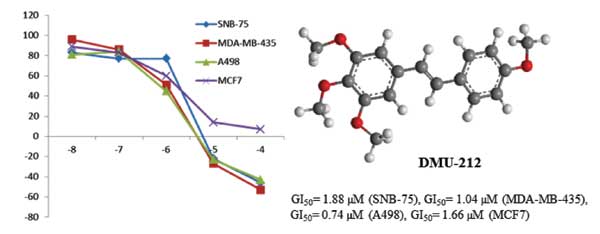
DMU-212, a methoxylated resveratrol analog, has significant anticancer activity, and selectively targets tumor cells. A library of E-diarylstilbenes structurally related to DMU-212 has been synthesized and evaluated for anticancer activity against a large panel of 45 human cancer cell lines. From this study, DMU-212 (3a) exhibited an average growth inhibitory effect (GI50) of 3.5 M against all the human cancer cell lines in the panel, and was particularly effective against the four cancer cell lines: SNB-75 (CNS), MDA-MB-435 (melanoma), A498 (renal), and MCF7 (breast), with GI50 values of 1.88, 1.04, 0.74 and 1.66 µM, respectively. Also, the 4’-chloro analog of DMU-212, 3d, exhibited 98 and 80 percent growth inhibition against MDA-MB-435 (melanoma) and K-562 (leukemia) cancer cell lines at a concentration of 10 M. Further investigation of DMU-212 and its analogs may provide novel therapeutic avenues for treatment of a variety of human cancers.
Keywords: Resveratrol, DMU-212, anticancer, growth inhibition, cytotoxicity, diarylstilbenes.